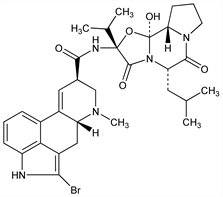
1416. Bromocriptine
Nomenclature
Description and references
Dopamine D2 receptor agonist; derivative of the ergotoxin group of ergot alkaloids. Prepn: E. Flückiger et al., DE 1926045; eidem, US 3752814 (1969, 1973 both to Sandoz). Pharmacology: E. Flückiger, H. R. Wagner, Experientia 24, 1130 (1968); E. Del Pozo et al., Schweiz. Med. Wochenschr. 103, 847 (1973). Relationship of stereochemistry and biological activity: H. P. Weber, Adv. Biochem. Psychopharmacol. 23, 25 (1980); N. Camerman, A. Camerman, Mol. Pharmacol. 19, 517 (1981). LC/MS/MS determn in plasma: A. Salvador et al., J. Chromatogr. B 820, 237 (2005). Long term clinical trial in Parkinson's disease: T. Nakanishi et al., Eur. Neurol. 32, Suppl. 1, 9 (1992). Clinical effect on body weight and glucose tolerance in obesity: A. H. Cincotta, A. H. Meier, Diabetes Care 19, 667 (1996). Clinical study in type 2 diabetes: H. Pijl et al., ibid. 23, 1154 (2000). Effects of prolactin concentration and estrus in beagles: N. J. Beijerink et al., Theriogenology 60, 1379 (2003). Comprehensive description: D. A. Giron-Forest, W. D. Schnleber, Anal. Profiles Drug Subs. 8, 47-81 (1979). Review of pharmacology, toxicology and therapeutic uses: D. Parkes, Adv. Drug Res. 12, 247-344 (1977); of therapeutic applications in endocrine and neurological diseases: K. Y. Ho, M. O. Thorner, Drugs 36, 67-82 (1988); of clinical experience in rheumatic and autoimmune diseases: R. W. McMurray, Semin. Arthritis Rheum. 31, 21-32 (2001).
Properties
Crystals from methyl ethyl ketone-isopropyl ether, mp 215-218° (dec). [α]D20 195° (c = 1 in methylene chloride).Derivative
Methanesulfonate.Nomenclature
Properties
Crystals from methyl ethyl ketone, mp 192-196° (dec). [α]D20 +95° (c = 1 in methanol-methylene chloride). Soly at 25° (mg/ml): methanol 910; ethanol 23.0; water 0.8; chloroform 0.45; benzene <0.1; hexane <0.1. pKa 4.90. LD50 in mice, rats, rabbits (mg/kg): 190, 72, 12.5 i.v. (Parkes).Therapeutic Category
Prolactin inhibitor; antiparkinsonian; antidiabetic.
Therapeutic Category (Veterinary)
Prolactin inhibitor.Keywords
Antiparkinsonian; Dopamine Receptor Agonist; Prolactin Inhibitor; Antidiabetic