1463. Bucladesine
Nomenclature
Description and references
Vasodilating cyclic nucleotide derivative which can permeate the cell membrane. Mimics the action of endogenous cyclic AMP, q.v. Prepn: Th. Posternak et al., Biochim. Biophys. Acta 65, 558 (1962); O. Nagase et al., JP Kokai 76 113896, C.A. 86, 140409r (1977); T. Hirayama et al., JP Kokai 77 39698; JP 77 39699, C.A. 87, 136330m-136331n (1977) (1976, 1977, 1977 all to Daiichi Seiyaku). Pharmacology: H. S. White et al., Eur. J. Pharmacol. 57, 107 (1979); J. D. Johnson et al., ibid. 91, 343 (1983); N. Bondar et al., J. Physiol. 355, 33 (1984). Clinical trial in congestive heart failure: S. Matsui et al., Am. J. Cardiol. 51, 1364 (1983); after cardiopulmonary bypass surgery: T. Yoshitake et al., Prog. Clin. Biol. Res. 111, 211 (1983).
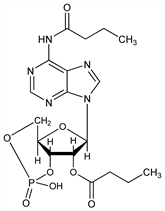
Derivative
Sodium salt.Nomenclature
Derivative
Barium salt.Nomenclature
Properties
uv max (ethanol-0.5M ammonium acetate, 5:2) 270 nm.Therapeutic Category
Cardiotonic.
Keywords
Cardiotonic