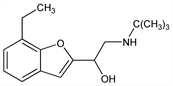
1480. Bufuralol
Nomenclature
Description and references
β-Adrenergic blocker with peripheral vasodilating activity. Prepn: NL 6606441; G. A. Fothergill et al., US 3929836 (1966, 1975 both to Hoffmann-La Roche). Prepn and resolution of isomers: eidem, Experientia 31, 1322 (1975); eidem, Arzneim.-Forsch. 27, 981 (1977). Pharmacology: T. C. Hamilton, M. W. Parkes, ibid. 1410. HPLC determn in plasma: P. Haefelfinger, J. Chromatogr. 221, 327 (1980). Metabolism of isomers and racemate: R. J. Francis et al., Eur. J. Clin. Pharmacol. 23, 529 (1982). Pharmacokinetics in hypertensive patients: M. Eckert et al., ibid. 24, 479 (1983). Hemodynamic effects in patients with angina: M. Pfisterer et al., J. Cardiovasc. Pharmacol. 6, 417 (1984).
Derivative
Hydrochloride.Nomenclature
Properties
Fine, white powder from acetone, mp 146°. LD50 in mice (mg/kg): 29.7 i.v.; 88.0 i.p.; 177 orally; in rats (mg/kg): 1400 s.c.; 750 orally (Hamilton, Parkes).Derivative
(+)-Hydrochloride.Nomenclature
Properties
Crystals from ethyl acetate-ether, mp 122-123°. [α]20365 +135.0° (c = 1.0 in ethanol).Derivative
()-Hydrochloride.Nomenclature
Properties
Crystals from ethyl acetate-ether, mp 122-123°. [α]20365 136.0° (c = 1.0 in ethanol).Therapeutic Category
Antianginal; antihypertensive.
Keywords
β-Adrenergic Blocker; Antianginal; Antihypertensive; Arylethanolamine Derivatives