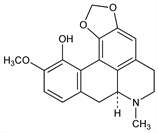
1481. Bulbocapnine
Nomenclature
CAS number: 298-45-3
(7aS)-6,7,7a,8-Tetrahydro-11-methoxy-7-methyl-5H-benzo[g]-1,3-benzodioxolo[6,5,4-de]quinolin-12-ol; 10-methoxy-1,2-(methylenedioxy)-6aα-aporphin-11-ol. C19H19NO4; mol wt 325.36.
C 70.14%, H 5.89%, N 4.30%, O 19.67%.
Description and references
From root of Corydalis cava (L.) Schweigg. & Krte (C. tuberosa DC), Fumariaceae and Dicentra canadensis Walp., Papaveraceae. Isoln: Freund, Josephi, Ber. 25, 2411 (1892); Manske, Can. J. Res. 7, 258 (1932). Pharmacology and toxicity study: H. Molitar, J. Pharmacol. Exp. Ther. 56, 85 (1936). Structure: Gadamer, Chem. Ztg. 34, 1004 (1910). Configuration: Ayer, Taylor, J. Chem. Soc. 1956, 472; Corrodi, Hardegger, Helv. Chim. Acta 39, 889 (1956). Synthesis: Kikkawa, C.A. 54, 4649b (1960). Peripheral-dopamine-receptor blocking activity: R. G. Pendleton et al., Arch. Pharmacol. 289, 171 (1975).
Derivative
dl-Form.Properties
Columns, mp 213-214°. Absorption max: Brustier, C.A. 49, 12127b (1955).Derivative
l-Form.Properties
Columns, mp 202-203°. [α]D22 239°.Derivative
d-Form.Properties
Columns, mp 201-203°. [α]D22 +231°. Practically insol in water, sol in alcohol, chloroform. LD50 s.c. in mice: 195 mg/kg (Molitar).Derivative
Methyl ether.C20H21NO4; mol wt 339.39.
C 70.78%, H 6.24%, N 4.13%, O 18.86%.