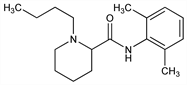
1495. Bupivacaine
Nomenclature
Description and references
Prepn: B. Ekenstam et al., Acta Chem. Scand. 11, 1183 (1957); B. T. Ekenstam, B. G. Pettersson, US 2955111 (1960 to AB Bofors). Resolution of isomers: B. F. Tullar, J. Med. Chem. 14, 891 (1971). Stereospecific synthesis: B. Adger et al., Tetrahedron Lett. 37, 6399 (1996). Pharmacology of racemate: F. Henn, R. Brattsand, Acta Anaesthesiol. Scand. Suppl. 21, 9 (1966), C.A. 66, 17863u (1967); of isomers: F. P. Luduena et al., Arch. Int. Pharmacodyn. 200, 359 (1972). Clinical pharmacokinetics: D. W. Blake et al., Anaesth. Intensive Care 22, 522 (1994). Comprehensive description: T. D. Wilson, Anal. Profiles Drug Subs. 19, 59-94 (1990). Review of use in spinal anesthesia: Acta Anaesthesiol. Scand. 35, 1-10 (1991). Review of pharmacology and clinical efficacy of levobupivacaine: K. J. McClellan, C. M. Spencer, Drugs 56, 355-362 (1998).
Properties
mp 107.5-108°. pKa 8.09; also reported as 8.17. Partition coefficient: (oleyl alcohol/water) 1565; (n-heptane/pH 7.4 buffer) 27.5.Derivative
Hydrochloride monohydrate.Nomenclature
Properties
White, odorless crystalline powder. mp 258.5°. Slightly sol in acetone, chloroform, ether. Soly (mg/ml): water 40; alcohol 125. LD50 in mice (mg/kg): 7.8 i.v., 82 s.c. (Henn, Brattsand).Derivative
(-)-Form.Nomenclature
Properties
Crystals from isopropanol, mp 135-137°. [α]D25 -80.9° (c = 5 in methanol).Derivative
(-)-Form hydrochloride.Nomenclature
Properties
mp 255-257°. [α]D25 -12.3° (c = 2 in water).Therapeutic Category
Anesthetic (local).
Keywords
Anesthetic (Local)