1497. Buprenorphine
Nomenclature
Description and references
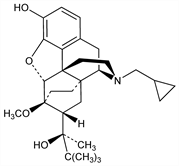
Semisynthetic derivative of thebaine, q.v. with mixed opioid agonist-antagonist properties. Prepn: K. W. Bentley, GB 1136214; idem, US 3433791 (1968, 1969 to Reckitt & Sons). See also: K. W. Bentley, “The Morphine Alkaloids” in The Alkaloids vol. 13, R. F. Manske, Ed. (Academic Press, New York, 1971) pp 75-120. Review of synthesis and analgesic activity: J. W. Lewis, “Ring C-Bridged Derivatives of Thebaine and Oripavine” in Advan. Biochem. Psychopharmacol. vol. 8, M. C. Braude et al., Eds. (Raven Press, New York, 1974) pp 123-137. NMR and stereochemistry: B. C. Uff et al., Magn. Reson. Chem. 23, 454 (1985). LC/ESI-MS/MS determn in urine, blood and hair: D. Favretto et al., Rapid Commun. Mass Spectrom. 20, 1257 (2006). Review of pharmacology and therapeutic efficacy in cancer pain: M. P. Davis, Support. Care Cancer 13, 878-887 (2005); in opioid addiction: S. E. Robinson, CNS Drugs 20, 697-712 (2006).
Properties
Crystals, mp 209°.Derivative
Hydrochloride.Nomenclature
Properties
White powder. Highly lipophilic. Slightly acidic with limited solubility in water.Note
This is a controlled substance (narcotic): 21 CFR, 1308.13.Therapeutic Category
Analgesic; in treatment of opioid dependence.
Keywords
Analgesic; Opioids; Oripavine Derivatives