1504. Buspirone
Nomenclature
Description and references
Non-benzodiazepine anxiolytic; 5-hydroxytryptamine (5-HT1) receptor agonist. Prepn: Y. H. Wu et al., J. Med. Chem. 15, 477 (1972); Y. H. Wu, J. W. Rayburn, DE 2057845 (1971 to Bristol-Myers); eidem, US 3717634 (1973 to Mead-Johnson). Pharmacology: L. E. Allen et al., Arzneim.-Forsch. 24, 917 (1974). Comparison with diazepam in treatment of anxiety: H. L. Goldberg, R. J. Finnerty, Am. J. Psychiatry 136, 1184 (1979). Disposition and metabolism: S. Caccia et al., Xenobiotica 13, 147 (1983). Series of articles on chemistry, pharmacology, addictive potential, and clinical trials: J. Clin. Psychiatry 43, pp 1-116 (1982); on pharmacology, safety and clinical comparison with clorazepate: Am. J. Med. 80, Suppl. 3B, 1-51 (1986). Review of pharmacology and therapeutic efficacy: K. L. Goa, A. Ward, Drugs 32, 114-129 (1986); M. W. Jann, Pharmacotherapy 8, 100-116 (1988); of discovery and clinical development: J. S. New, Med. Res. Rev. 10, 283-326 (1990); of pharmacokinetics and pharmacodynamics: I. Mahmood, C. Sahajwalla, Clin. Pharmacokinet. 36, 277-287 (1999).
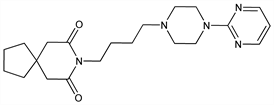
Derivative
Hydrochloride.Nomenclature
Properties
Crystals from abs ethanol, mp 201.5-202.5°. Highly sol in water. pKa1: 4.12. pKa2: 7.32. LD50 i.p. in rats: 136 mg/kg (Allen).Therapeutic Category
Anxiolytic.
Therapeutic Category (Veterinary)
In treatment of behavioral disorders in dogs and cats.Keywords
Anxiolytic; Arylpiperazines; Serotonin Receptor Agonist