1505. Busulfan
Nomenclature
Description and references
Cytotoxic alkylating agent with antileukemic activity; conditioning agent in bone marrow transplants. Discovery: A. Haddow, G. M. Timmis, Lancet 1, 207 (1953). Prepn: G. M. Timmis, US 2917432 (1959 to Burroughs Wellcome). Comprehensive description: M. Tariq, A. A. Al Badr, Anal. Profiles Drug Subs. 16, 53-83 (1987). Pharmacokinetics: H. Ehrsson et al., Clin. Pharmacol. Ther. 34, 86 (1983). Review of pharmacology: C. D. R. Dunn, Exp. Hematol. 2, 101-117 (1974); of toxicology: J. B. Bishop, J. S. Wassom, Mutat. Res. 168, 15-45 (1986). Chemosterilant effect in boll weevils: J. W. Haynes et al., J. Econ. Entomol. 66, 619 (1973). LC/MS determn in plasma and saliva: M. Rauh et al., Clin. Pharmacokinet. 45, 305 (2006). Clinical pretreatment with cyclophosphamide for bone marrow transplants: G. W. Santos et al., N. Engl. J. Med. 309, 1347 (1983). Clinical pharmacokinetics in stem cell transplant patients: L. Nguyen et al., Cancer Chemother. Pharmacol. 57, 191 (2006); in bone marrow transplantation in thalassemia: M. Chandy et al., Bone Marrow Transplant. 36, 839 (2005).
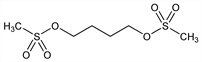
Properties
Crystals, mp 114-118°. Soly in acetone at 25°: 2.4 g/100 ml; in alcohol: 0.1 g/100 ml. Practically insol in water, but will dissolve slowly as hydrolysis takes place. LD50 i.v. in rats: 1.8 mg/kg. See: H. R. Scherf et al., Arzneim.-Forsch. 20, 1467 (1970).Caution
This substance is listed as a known human carcinogen: Report on Carcinogens, Eleventh Edition (PB2005-104914, 2004) p III-39.Use
Insect sterilant.Therapeutic Category
Antineoplastic.
Therapeutic Category (Veterinary)
Antineoplastic.Keywords
Antineoplastic; Alkylating Agents; Alkyl Sulfonates