1507. Butacaine
Nomenclature
CAS number: 149-16-6
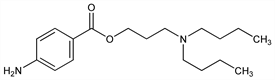
3-(Dibutylamino)-1-propanol 4-aminobenzoate; 3-(p-aminobenzoxy)-1-di-n-butylaminopropane; dibutylaminopropyl-p-aminobenzoate; p-aminobenzoyldibutylaminopropanol; Butelline. C18H30N2O2; mol wt 306.44.
C 70.55%, H 9.87%, N 9.14%, O 10.44%.
Description and references
Prepd from p-nitrobenzoyl chloride and γ-di-n-butylaminopropanol followed by reduction of the NO2 group to NH2: O. Kamm et al., US 1358751; Adams, Volwiler, US 1676470; A. Weston, US 2437984 (1920, 1928, 1948 all to Abbott); Burnett et al., J. Am. Chem. Soc. 59, 2248 (1937); Kaye, Roberts, ibid. 73, 4762 (1951). Toxicity study: Schmidt et al., Toxicol. Appl. Pharmacol. 1, 454 (1956).
Properties
Liquid, bp0.11 178-182°.Derivative
Sulfate.Nomenclature
CAS number: 149-15-5
Butyn Sulfate (Abbott). (C18H30N2O2)2.H2SO4; mol wt 710.96.
C 60.82%, H 8.79%, N 7.88%, O 18.00%, S 4.51%.
Properties
Crystals from n-propanol, mp 138.5-139.5°. Also reported as mp 100-103°. Produces numbness of tongue on tasting. Affected by light. One gram dissolves slowly in somewhat less than 1 ml water, more rapidly on heating. Quite sol in warm alc, in acetone; slightly sol in chloroform. Practically insol in ether. Aq soln is practically neutral to litmus and may be boiled for sterilization without dec. LD50 i.v. in mice: 12.4 mg/kg (Schmidt). Pharmaceutical Incompat. Alkalies and alkaline-reacting substances liberate the free base as an oily liquid from solns. Bicarbonates produce a precipitate of butacaine carbonate. Iodine gives a brown precipitate. Chlorides form the almost insol butacaine chloride which may precipitate.Derivative
Hydrochloride.C18H30N2O2.HCl; mol wt 342.90.
C 63.05%, H 9.11%, N 8.17%, O 9.33%, Cl 10.34%.
Properties
Crystals from ethanol, mp 157-158.5°.Therapeutic Category
Anesthetic (local).
Therapeutic Category (Veterinary)
Anesthetic (local).Keywords
Anesthetic (Local)