Nomenclature
CAS number: 107-88-0
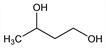
1,3-Butanediol; 1,3-dihydroxybutane; β-butyleneglycol; methyltrimethylene glycol; butane-1,3-diol.
C
4H
10O
2; mol wt 90.12.
C 53.31%, H 11.18%, O 35.51%.
Description and references
Usually prepd by catalytic hydrogenation of
aldol using Raney nickel: Hancock, Henson, Ind. Eng. Chem. 45, 629 (1953); GB 853266 (1960 to Celanese); F. S. Wagner in Kirk-Othmer Encyclopedia
of Chemical Technology vol. 11 (Wiley-Interscience,
New York, 3rd ed., 1980) pp 956-962. Toxicity: Smyth et al., Arch. Ind. Hyg. Occup. Med. 4, 119 (1951).
Properties
Viscous liquid. The pure compd is colorless.
d2020 1.004-1.006. One gallon weighs 8.398 lbs at room temp. Viscosity (cSt):
24.6 (50°); 96 (25°); 590 (0°); 3253 (-17.7°); 6059 (-23°). mp < 50°. Because
of the high viscosity at low temps heating is necessary for pumping.
Very hygroscopic: Will absorb 38.5 wt % of water within 144 hrs.
at 81% relative humidity. bp 207.5°. nD20 1.4401. Flash pt (tag open cup): 250°F (121°C). Dielectric constant 28.8 at 25°. Surface
tension: 37.8 dynes/cm at 25°. Sol in water, acetone, methyl ethyl
ketone, ethanol, dibutyl phthalate, castor oil. Practically insol
in aliphatic hydrocarbons, benzene, toluene, carbon tetrachloride,
ethanolamines, mineral oil, linseed oil. LD50 orally in rats: 22.8 g/kg (Smyth).Use
Intermediate in the manufacture of polyester plasticizers,
humectant for cellophane, tobacco. Has some mold inhibiting action.
