1568. 2,3-Butylene Glycol
Nomenclature
Description and references
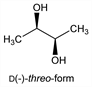
Occurs in 3 isomeric forms: meso- or erythro-, d(-)-threo-, and l(+)-threo-forms. The commercial product is usually either the meso- or the d(-)-form. Prepn of meso-form from trans-2,3-epoxybutane and of dl-form from cis-2,3-epoxybutane: Wilson, Lucas, J. Am. Chem. Soc. 58, 2396 (1936). Prepn of d(-)- and l(+)-forms from corresponding d- and l-mannitols: Rubin et al., ibid. 74, 425 (1952). Manuf of d(-)-form by fermentation of carbohydrate solns with organisms of the Bacillus subtilis group: Vergnaud, US 2529061 (1950 to Usines de Melle). Manuf from 2-butene: Cosby et al., US 2808429 (1957 to Allied Chem.); Keith et al., US 2974161 (1961 to Sinclair); from 2-butyne: Saegebarth, US 3157704 (1964 to du Pont). Configuration: Morell, Auernheimer, J. Am. Chem. Soc. 66, 792 (1944); Leroux, Lucas, ibid. 73, 41 (1951); Rubin et al., loc. cit.