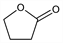
1596. Butyrolactone
Nomenclature
CAS number: 96-48-0
Dihydro-2(3H)-furanone; γ-butyrolactone; 1,2-butanolide; 1,4-butanolide; γ-hydroxybutyric acid lactone; 3-hydroxybutyric acid lactone; 4-hydroxybutanoic acid lactone. C4H6O2; mol wt 86.09.
C 55.81%, H 7.02%, O 37.17%.
Description and references
Prepd from acetylene and formaldehyde: Reppe, Chem. Ing. Tech. 1950, 365; Chem. Eng. 58, no. 6, 176 (1951); also prepd from ethylene chlorohydrin, glutaric acid, ν-hydroxybutyric acid solns, tetrahydrofuran, or vinylacetic acid: F. C. Whitmore, Organic Chemistry (Van Nostrand, New York, 2nd ed., 1951). Alternate synthesis: Y. Ogata et al., J. Org. Chem. 45, 1320 (1980). Physical properties: McKinley, Copes J. Am. Chem. Soc. 72, 5331 (1950). Toxicity: H. F. Smyth et al., Am. Ind. Hyg. Assoc. J. 30, 470 (1969).