1606. Cactinomycin
Nomenclature
Description and references
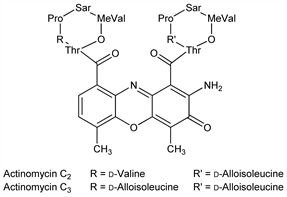
Antibiotic complex produced by Streptomyces chrysomallus: Brockmann, Grubhofer, Naturwissenschaften 36, 376 (1949); 37, 494 (1950); Ber. 84, 260 (1951); US 2953495 (1960 to Shenley Inds.); Lindenbein, Arch. Mikrobiol. 17, 361 (1952). Mixture of actinomycins C1 (dactinomycin, q.v.), C2 and C3, 10%, 45% and 45%, resp: Brockmann, Pfennig, Naturwissenschaften 39, 429 (1952); Brockmann, Gr"one, ibid. 40, 222 (1953). Description of other actinomycins: Waksman et al., Proc. Natl. Acad. Sci. USA 44, 602 (1958). Structures: Brockmann et al., Angew. Chem. 68, 70 (1956); Brockmann, Boldt, Naturwissenschaften 50, 19 (1963). Synthesis of actinomycin C3: Brockmann, Lackner, ibid. 47, 230 (1960); 48, 555 (1961); 51, 407 (1964); Brockmann et al., DE 1172680 (1964 to Bayer); Brockmann, Lackner, Ber. 100, 353 (1967); 101, 1312 (1968). Synthesis of actinomycin C2: Brockmann, Lackner, Tetrahedron Lett. 1964, 3517. Comprehensive review: H. Brockmann, in Fortschr. Chem. Org. Naturst. 18, 1-54 (1960).
Properties
Alizarin-red hexagonal bipyramids from ethyl acetate, mp 252°. [α]D25 -325 to -349° (c = 0.25 in ethanol). Sparingly sol in water; moderately sol in ethanol; sol in chloroform, ethyl acetate, benzene, acetone. Protect from light.Derivative
Actinomycin C2.Properties
Red bipyramids, prisms or needles from ethyl acetate, mp 237-239°. [α]D21 -325 ±10° (c = 0.23 in methanol). Abs max (methanol): 443 nm (ε 25400).Derivative
Actinomycin C3.Properties
Red hexagonal bipyramids from ethyl acetate or methanol, dec 235°. [α]D17 -328° (c = 0.5 in ethanol). Absorption max (methanol): 443 nm (ε 24100). Weak base.Therapeutic Category
Antineoplastic.
Keywords
Antineoplastic; Antibiotics and Analogs; Actinomycins