1638. Calcifediol
Nomenclature
Description and references
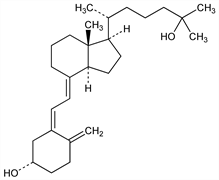
The principal circulating form of vitamin D3, formed in the liver by hydroxylation at C-25: Ponchon, DeLuca, J. Clin. Invest. 48, 1273 (1969). It is the intermediate in the formation of 1α,25-dihydroxycholecalciferol, q.v., the biologically active form of vitamin D3 in the intestine. Identification in rat as an active metabolite of vitamin D3: Lund, DeLuca, J. Lipid Res. 7, 739 (1966); Morii et al., Arch. Biochem. Biophys. 120, 513 (1967). Evaluation of biological activity in comparison with vitamin D3: Blunt et al., Proc. Natl. Acad. Sci. USA 61, 717 (1968); ibid. 1503. Isoln from porcine plasma and establishment of structure: Blunt et al., Biochemistry 7, 3317 (1968). Synthesis: Blunt, DeLuca, ibid. 8, 671 (1969). Review of isoln, identification and synthesis: DeLuca, Am. J. Clin. Nutr. 22, 412 (1969). Review of bioassays: J. G. Haddad Jr., Basic Clin. Nutr. 2, 579-597 (1980).
Properties
uv max (ethanol): 265 nm (ε 18000) (Blunt, DeLuca).Therapeutic Category
Calcium regulator.
Keywords
Calcium Regulator