1639. Calcimycin
Nomenclature
Description and references
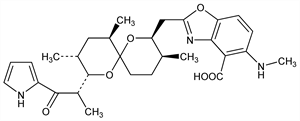
Polyether antibiotic produced by a strain of Streptomyces chartreusensis Calhoun and Johnson NRRL 3882. Activity as a divalent cation ionophore in isolated mitochondria: P. W. Reed, H. A. Lardy, J. Biol. Chem. 247, 6970 (1972). Prepn and antimicrobial activity: R. M. Gale et al., US 3923823 (1975 to Lilly). Elucidation of structure: M. O. Chaney et al., J. Am. Chem. Soc. 96, 1932 (1974). Spectral studies of ionophore and metal ion complexes: D. R. Pfeiffer et al., Biochemistry 13, 4007 (1974). Total synthesis and absolute configuration: D. A. Evans et al., J. Am. Chem. Soc. 101, 6789 (1979); P. A. Grieco et al., J. Org. Chem. 45, 3537 (1980). Stereospecific synthesis: G. R. Martinez et al., J. Am. Chem. Soc. 104, 1436 (1982); D. P. Negri, Y. Kishi, Tetrahedron Lett. 28, 1063 (1987). Review of cation binding and transport properties: D. R. Pfeiffer et al., Ann. N.Y. Acad. Sci. 307, 402-423 (1978). Use in model systems of calcium transport: M. Takamori et al., J. Neurol. Sci. 50, 89 (1981); M. Takamori et al., ibid. 51, 207 (1981); M. H. Freedman et al., Cell. Immunol. 58, 134 (1981); G. Thomas, Eur. J. Pharmacol. 81, 35 (1982); V. L. Lew, J. Garcia-Sancho, Cell Calcium 6, 15 (1985).