1752. Cantharidin
Nomenclature
CAS number: 56-25-7
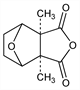
Hexahydro-3a,7a-dimethyl-4,7-epoxyisobenzofuran-1,3-dione; 2,3-dimethyl-7-oxabicyclo[2.2.1]heptane-2,3-dicarboxylic
anhydride; exo-1,2-cis-dimethyl-3,6-epoxyhexahydrophthalic
anhydride; cantharides camphor. C10H12O4; mol wt 196.20.
C 61.22%, H 6.16%, O 32.62%.
Description and references
Defensive toxin produced by blister beetles of the family Meloidae. Active principle of cantharides, q.v. and notorious “Spanish Fly” aphrodisiac. Synthesis and stereochemistry: Woodward, Loftfield, J. Am. Chem. Soc. 63, 3167 (1941); Ziegler et al., Ann. 551, 1 (1942). Stereospecific synthesis: Stork, Van Tamelen et al., J. Am. Chem. Soc. 75, 384 (1953). Simple efficient synthesis: W. G. Dauben et al., ibid. 102, 6893 (1980). Crystal structure: M. Zehnder, U. Thewalt, Helv. Chim. Acta 60, 740 (1977). Review of biological source and medicinal uses: L. Moed et al., Arch. Dermatol. 137, 1357-1360 (2001).
Properties
Orthorhombic plates, scales, mp 218°. Sublimes at about 110° (12 mm Hg, 3-5 mm distance). Insol in cold water, somewhat sol in hot water. One gram dissolves in 40 ml acetone, 65 ml chloroform, 560 ml ether, 150 ml ethyl acetate. Sol in oils.Caution
Extreme irritant and vesicant. Direct contact may cause intense blister formation. Potential symptoms of overexposure are burning sensation of throat and oral cavity; diarrhea, hemorrhagic necrosis of upper GI tract, abdominal pain; urinary urgency, strangury, hematuria, priapism, oliguria, tubular necrosis, renal failure. See Clinical Toxicology of Commercial Products, R. E. Gosselin et al., Eds. (Williams & Wilkins, Baltimore, 5th ed., 1984) Section II, 270.Therapeutic Category
Vesicant; in treatment of warts and molluscum.