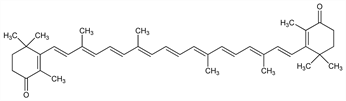
1753. Canthaxanthin
Nomenclature
CAS number: 514-78-3
β,β-Carotene-4,4′-dione; 4,4′-dioxo-β-carotene; Food Orange 8; C.I. 40850; Carophyll Red (Roche); Orobronze (Applipharm); Roxanthin Red 10 (Roche); Carotaben plus. C40H52O2; mol wt 564.84.
C 85.06%, H 9.28%, O 5.67%.
Description and references
All trans-carotenoid pigment widely distributed in nature. Isoln from the edible mushroom Cantharellus cinnabarinus Adans. ex Fr., Agaricaceae: Haxo, Bot. Gaz. 112, 228 (1950); also isolated from flamingo feathers. Structure and synthesis: Isler et al., Verh. Naturforsch. Ges. Basel 67, 379 (1956); Zeller et al., Helv. Chim. Acta 42, 841 (1959). Alternate syntheses: M. Akhtar, B. C. L. Weedon, J. Chem. Soc. 1959, 4058; R. Rüegg, G. Saucy, US 2983752 and J. D. Surmatis, US 3311656 (1961, 1967 both to Hoffmann-La Roche); J. D. Surmatis et al., Helv. Chim. Acta 53, 974 (1970); M. Rosenberger et al., Pure Appl. Chem. 51, 871 (1979); eidem, J. Org. Chem. 47, 2130 (1982).