1774. Captopril
Nomenclature
Description and references
First orally active angiotensin-converting enzyme (ACE) inhibitor. Prepn: M. A. Ondetti, D. W. Cushman, DE 2703828; eidem, US 4046889 and US 4105776 (1977, 1977, 1978 all to Squibb). Design and synthesis: M. A. Ondetti et al., Science 196, 441 (1977) DOI PubMed; D. W. Cushman et al., Biochemistry 16, 5484 (1977) DOI PubMed. Improved synthesis: D. H. Nam et al., J. Pharm. Sci. 73, 1843 (1984) DOI PubMed. Pharmacology: B. Rubin et al., Eur. J. Pharmacol. 51, 377 (1978) DOI PubMed; eidem, Prog. Cardiovasc. Dis. 21, 183 (1978) DOI PubMed. Clinical studies: D. B. Case et al., ibid. 195 DOI PubMed; H. R. Brunner et al., Ann. Intern. Med. 90, 19 (1979) PubMed. Toxicology and metabolism: G. R. Keim in Captopril and Hypertension, D. B. Case, Ed. (Plenum, New York, 1980) p 137. GC/MS determn in biological fluids: T. Ito, Y. Matsuki, J. Chromatogr. 417, 79 (1987) DOI PubMed. HPLC determn in pharmaceutical formulations: T. Mirza, H. S. I. Tan, J. Pharm. Biomed. Anal. 25, 39 (2001) DOI PubMed. Thermal analysis: Y. Huang et al., Thermochim. Acta 367-368, 43 (2001) DOI. Historical review and comprehensive bibliography: Z. P. Horovitz in Pharmacological and Biochemical Properties of Drug Substances vol. 3, M. E. Goldberg, Ed. (Am. Pharm. Assoc., Washington, DC, 1981) pp 148-175. Comprehensive description: H. Kadin, Anal. Profiles Drug Subs. 11, 79-137 (1982). Reviews: Am. Heart J. 104, 1125-1228 (1982) DOI; Br. J. Clin. Pharmacol. 14, Suppl. 2, 69S-252S (1982). Series of articles on pharmacology and therapeutic efficacy: Postgrad. Med. J. 62, Suppl. 1, 1-191 (1986).
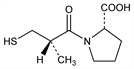
Properties
Crystals from ethyl acetate/hexane, mp 103-104° (Ondetti, Cushman). Generally regarded as polymorphic: stable form, mp 106°; unstable form, mp 86° (Florey); also reported as mp 87-88°, resolidifies, second mp 104-105° (Cushman). Slight sulfurous odor. [α]D22 -131.0° (c = 1.7 in ethanol). pK1 3.7, pK2 9.8. Freely sol in water (≈160mg/ml), methylene chloride; sol in methanol, ethanol; sparingly sol in chloroform, ethyl acetate. LD50 in mice (mg/kg): 1040 i.v.; 6000 orally (Keim).Therapeutic Category
Antihypertensive.
Therapeutic Category (Veterinary)
Antihypertensive.Keywords
ACE-Inhibitor; Antihypertensive; N-Carboxyalkyl (peptide/lactam) Derivatives