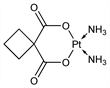
1822. Carboplatin
Nomenclature
Description and references
Analog of cisplatin, q.v., with reduced nephrotoxicity. Prepn and antitumor activity: NL 7307863; M. J. Cleare et al., US 4140707 (1973, 1979 both to Research Corp.). Improved prepn: R. C. Harrison et al., Inorg. Chim. Acta 46, L15 (1980). Crystal structure: S. Neidle et al., J. Inorg. Biochem. 13, 205 (1980). Comparison with other antitumor platinum complexes: M. J. Cleare et al., Biochimie 60, 835 (1978). Early clinical studies: A. H. Calvert et al., Cancer Chemother. Pharmacol. 9, 140 (1982). Clinical pharmacokinetics: S. J. Harland et al., Cancer Res. 44, 1693 (1984); M. J. Egorin et al., ibid. 5432. Toxicity, activity in mice, rats, dogs: P. Lelieveld et al., Eur. J. Cancer Clin. Oncol. 20, 1087 (1984). Comparison with cisplatin chemotherapy in advanced seminoma: M. J. Peckham et al., Br. J. Cancer 52, 7 (1985). In treatment of small cell lung cancer: I. E. Smith et al., Cancer Treat. Rep. 69, 43 (1985); in metastatic breast cancer: E. A. Perez, Oncologist 9, 518 (2004). Clinical pharmacokinetics: S. B. Duffull, B. A. Robinson, Clin. Pharmacokinet. 33, 161 (1997). Review of veterinary studies: L. E. Fox, J. Am. Anim. Hosp. Assoc. 36, 13-14 (2000).
Properties
White crystals, sol in water. LD50 in mice (mg/kg): 150 i.p., 140 i.v.; in rats (mg/kg): 85 i.v. (Lelieveld).Therapeutic Category
Antineoplastic.
Therapeutic Category (Veterinary)
Antineoplastic.Keywords
Antineoplastic; Platinum Complexes