1823. Carboprost
Nomenclature
CAS number: 35700-23-3
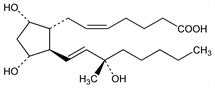
(5Z,9α,11α,13E,15S)-9,11,15-Trihydroxy-15-methylprosta-5,13-dien-1-oic
acid; 7-[3,5-dihydroxy-2-(3-hydroxy-3-methyl-1-octenyl)cyclopentyl]-5-heptenoic
acid; (15S)-15-methyl PGF2α; U-32921. C21H36O5; mol wt 368.51.
C 68.44%, H 9.85%, O 21.71%.
Description and references
Analog of prostaglandin F2α, q.v. Prepn: G. L. Bundy et al., DE 2121980; G. L. Bundy, US 3728382 (1971, 1973 both to Upjohn); eidem, Ann. N.Y. Acad. Sci. 180, 76 (1971); E. W. Yankee et al., J. Am. Chem. Soc. 96, 5865 (1974). Biological activity: J. R. Weeks et al., J. Pharmacol. Exp. Ther. 186, 67 (1973). Mechanism of action: A. I. Csapo, M. O. Pulkkinen, Prostaglandins 18, 479 (1979). Clinical studies: P. C. Schwallie, K. R. Lamborn, J. Reprod. Med. 23, 289 (1979); M. P. Mapa et al., Int. J. Gynaecol. Obstet. 20, 125 (1982). Teratological study: G. M. Szczech et al., Adv. Prostaglandin Thromboxane Res. 4, 157 (1978).
Derivative
Tromethamine salt.Nomenclature
CAS number: 58551-69-2
Carboprost trometamol; U-32921E; Hemabate (Pfizer); Prostin/15M (Pfizer). C25H47NO8; mol wt 489.64.
C 61.32%, H 9.68%, N 2.86%, O 26.14%.
Derivative
Methyl ester.Nomenclature
CAS number: 35700-21-1
Carboprost methyl; U-36384. C22H38O5; mol wt 382.53.
C 69.08%, H 10.01%, O 20.91%.
Properties
Crystals from ether/hexane, mp 55-56°. [α]D +24° (c = 0.81 in ethanol).Therapeutic Category
Oxytocic.
Keywords
Oxytocic; Prostaglandin/Prostaglandin Analog