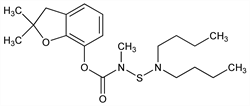
1826. Carbosulfan
Nomenclature
CAS number: 55285-14-8
[(Dibutylamino)thio]methylcarbamic acid 2,3-dihydro-2,2-dimethyl-7-benzofuranyl
ester; FMC-35001; Marshal (FMC); Posse (FMC). C20H32N2O3S; mol wt 380.54.
C 63.12%, H 8.48%, N 7.36%, O 12.61%, S 8.43%.
Description and references
Carbamate cholinesterase inhibitor which is hydrolyzed to carbofuran in soil. Prepn: A. L. Black, T. R. Fukuto, BE 817517; eidem, US 4006231 (1975, 1977 both to Regents Univ. Calif.). Metabolism in orange tree leaves and fruit: V. E. Clay, T. R. Fukuto, Arch. Environ. Contam. Toxicol. 13, 53 (1984). Degradation and persistence in soil: A. Sahoo et al., Bull. Environ. Contam. Toxicol. 50, 29 (1993). HPLC determn in oranges: M.W. Brooks, A. Barros, Analyst 120, 2479 (1995). Spectrophotometric determn in formulations and water samples: K. R. Mohan et al., Asian J. Chem. 10, 457 (1998). Review of physical properties and field trials: E. G. Maitlen, N. A. Sladen, Proc. Br. Crop Prot. Conf. - Pests Dis. 1979, 557-564.