1840. Carindacillin
Nomenclature
CAS number: 35531-88-5
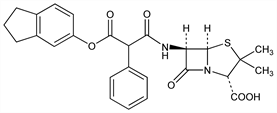
(2S,5R,6R)-6-[[3-[(2,3-Dihydro-1H-inden-5-yl)oxy]-1,3-dioxo-2-phenylpropyl]amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic
acid; N-(2-carboxy-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]hept-6-yl)-2-phenylmalonamic
acid 1-(5-indanyl) ester; 6-[2-(5-indanyloxycarbonyl)phenylacetamido]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic
acid; 1-(5-indanyl) N-(2-carboxy-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]hept-6-yl)-2-phenylmalonamate; 6-[2-phenyl-2-(5-indanyloxycarbonyl)acetamido]penicillanic
acid; 6-(2-carboxy-2-phenylacetamido)-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic
acid 6-(5-indanyl ester); α-(5-indanyloxycarbonyl)benzylpenicillin; carbenicillin indanyl ester; CP-15464. C26H26N2O6S; mol wt 494.56.
C 63.14%, H 5.30%, N 5.66%, O 19.41%, S 6.48%.
Description and references
Semi-synthetic antibiotic related to penicillin. Prepn: K. Butler, ZA 6900060; idem, US 3679801 (1969, 1972 both to Pfizer). New process: S. Nakanishi, US 3759898 (1973 to Pfizer). Activity studies: Butler, Del. Med. J. 43, 366 (1971); English et al., Antimicrob. Agents Chemother. 1, 185 (1972). Clinical evaluation: Wallace et al., Antimicrob. Agents Chemother. 1970, 223; Bran et al., Clin. Pharmacol. Ther. 12, 525 (1971); Indanyl Carbenicillin, H. Swarz, F. E. Storari, Eds. (Am. Elsevier, New York, 1974) 100 pp.
Derivative
Sodium salt.Nomenclature
CAS number: 26605-69-6
CP-15464-2; Carindapen (Pfizer); Geocillin (Roerig); G.U.-Pen (Pfizer). C26H25N2NaO6S; mol wt 516.54.
C 60.46%, H 4.88%, N 5.42%, Na 4.45%, O 18.58%, S 6.21%.
Properties
mp 207-213°.Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Penicillins