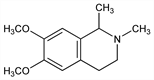
1847. Carnegine
Nomenclature
CAS number: 490-53-9
1,2,3,4-Tetrahydro-6,7-dimethoxy-1,2-dimethylisoquinoline; 6,7-dimethoxy-1,2-dimethyl-1,2,3,4-tetrahydroisoquinoline; pectenine. C13H19NO2; mol wt 221.30.
C 70.56%, H 8.65%, N 6.33%, O 14.46%.
Description and references
In Carnegiea gigantea (Engelm.) Britt. & Rose (Cereus giganteus Engelm.), Cactaceae, a cactus of Arizona and Mexico. Extraction procedure: Heyl, Arch. Pharm. 266, 668 (1928). Synthesis: Sp"ath, Ber. 62, 1021 (1929); Nakada, Nisgihara, J. Pharm. Soc. Jpn. 64, 74 (1944). Pharmacology: E. Santi-Soncin, M. Furlanut, Fitoterapia 43, 21 (1972), C.A. 80, 66667f (1974). Review of synthetic methods: A. B. J. Bracca, T. S. Kaufman, Tetrahedron 60, 10575-10610 (2004).
Properties
Viscous liquid, dec on standing. Distills at 1 mm pressure and 170° air bath temp. Sol in ether, alc, chloroform. LD50 i.p. in mice: 15.23 mg/kg (Santi-Soncin, Furlanut).Derivative
Hydrochloride monohydrate.Nomenclature
CAS number: 5864-18-6
C13H19NO2.HCl.H2O; mol wt 275.77.
C 56.62%, H 8.04%, N 5.08%, O 17.41%, Cl 12.86%.
Properties
Clusters from dil alc, mp 207° (mp 211° when anhydr). Sol in water, slightly sol in alc.Derivative
Hydrobromide monohydrate.Nomenclature
CAS number: 5853-25-8
C13H19NO2.HBr.H2O; mol wt 320.22.
C 48.76%, H 6.92%, N 4.37%, O 14.99%, Br 24.95%.
Properties
Needles from alc, mp 228°.Derivative
Methyliodide.Nomenclature
CAS number: 5911-58-0
C13H19NO2.CH3I; mol wt 363.23.
C 46.29%, H 6.10%, N 3.86%, O 8.81%, I 34.94%.