1871. Carumonam
Nomenclature
Description and references
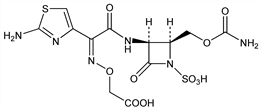
Synthetic monocyclic β-lactam (monobactam) antibiotic. Prepn: S. Kishimoto et al., EP 93376; T. Matsuo et al., US 4572801 (1983, 1986 both to Takeda); M. Sendai et al., J. Antibiot. 38, 346 (1985). Alternate synthesis: P. S. Manchard et al., J. Org. Chem. 53, 5507 (1988). Comparative in vitro antimicrobial activity: R. J. Fass, V. L. Helsel, Antimicrob. Agents Chemother. 28, 834 (1985); B. R. Smith et al., ibid. 29, 346 (1986); I. M. Hoepelman et al., Chemotherapy (Basel) 33, 103 (1987). β-Lactamase stability: R. L. Then, ibid. 30, 398 (1984). Pharmacokinetics in humans: E. Weidekamm et al., Antimicrob. Agents Chemother. 26, 898 (1984); C. A. M. McNulty et al., ibid. 28, 425 (1985).
Properties
Colorless powder. [α]D26 -45° (c = 1 in DMSO).Derivative
Disodium salt.Nomenclature
Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Monobactams