1872. Carvacrol
Nomenclature
CAS number: 499-75-2
2-Methyl-5-(1-methylethyl)phenol; 2-p-cymenol; 2-hydroxy-p-cymene; isopropyl-o-cresol; isothymol. C10H14O; mol wt 150.22.
C 79.95%, H 9.39%, O 10.65%.
Description and references
Found in oil of origanum, thyme, marjoram, summer savory: E. Guenther, The Essential Oils vol. 2 (Van Nostrand, New York, 1949) p 503; Carpenter, Easter, J. Org. Chem. 20, 401 (1955). Prepn by chlorination of α-pinene with tert-butyl hypochlorite: Ritter, Ginsburg, J. Am. Chem. Soc. 72, 2381 (1950); from 2-bromo-p-cymol: Strubell, Baumgartel, Arch. Pharm. 291, 66 (1958). Toxicity data: Kochmann, Arch. Exp. Pathol. Pharmakol. 161, 196 (1931).
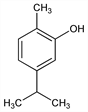
Properties
Liquid; thymol odor. d420 0.976; d2525 0.9751. bp760 237-238°; bp18 118-122°; bp3 93°. mp ≈0°. nD20 1.52295. uv max (95% ethanol): 277.5 nm (log ε 3.262). Volatile with steam. Practically insol in water. Freely soluble in alc or ether. LD orally in rabbits: 100 mg/kg (Kochmann).Use
As disinfectant; in organic syntheses.Therapeutic Category
Has been used as anti-infective; anthelmintic (Nematodes).
Keywords
Anthelmintic (Nematodes); Antiseptic/Disinfectant; Phenols