1902. Catechin
Nomenclature
Description and references
Flavonoid found primarily in higher woody plants as (+)-catechin along with (-)-epicatechin (cis form). From catechu (gambir and acacia), mahogany wood, etc.: Perkin, Yoshitake, J. Chem. Soc. 81, 1160 (1902); Freudenberg et al., Ber. 55, 1734 (1922). Structure: Freudenberg et al., Ann. 444, 135 (1925). Stereochemistry: Clark-Lewis, Chem. Ind. (London) 1955, 1218; Hardegger et al., Helv. Chim. Acta 40, 1819 (1957); Clark-Lewis, J. Chem. Soc. 1960, 2433. Pharmacology: Van Cauwenberge et al., C. R. Seances Soc. Biol. Ses Fil. 165, 1195 (1971). Metabolism: Das, Sothy, Biochem. J. 125, 417 (1971); and absorption in human: N. P. Das, Biochem. Pharmacol. 20, 3435 (1971). Biosynthesis of epicatechins: Zaprometov, Grisebach, Z. Naturforsch. 28c, 113 (1973). Other catechins such as afzelechin and gallocatechin and catechin oligomers (procyanidins) also exist in nature: Thompson et al., J. Chem. Soc. Perkin Trans. 1 1972, 1287. See also Bioflavonoids.
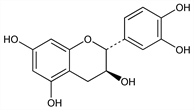
Properties
Needles from water + acetic acid, occurs as hydrate. mp 93-96°; 175-177° when anhydr. [α]D18 +16 to +18.4°.Derivative
dl-Form.Nomenclature
Properties
Needles from water + acetic acid, mp 212-216°. Slightly sol in cold water, ether; sol in hot water, alcohol, glacial acetic acid, acetone. Practically insol in benzene, chloroform, petr ether.Derivative
l-Form.Nomenclature
Properties
Needles from water + acetic acid, mp 93-96°; 175-177° when anhydr. [α]D -16.8°.Note
Catechin is also called catechol (flavan) to distinguish it from catechol (pyrocatechol, q.v.).Use
In dyeing and tanning.Therapeutic Category
Antidiarrheal.
Keywords
Antidiarrheal; Hepatoprotectant