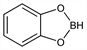
10309. Catecholborane
Nomenclature
Description and references
Versatile boron hydride reagent used in synthesis to perform hydroborations and reductions. Prepn from 2-chloro-1,3,2-benzodioxaborole: H. C. Newsom, W. G. Woods, Inorg. Chem. 7, 177 (1968) DOI; from catechol: H. C. Brown, S. K. Gupta, J. Am. Chem. Soc. 93, 1816 (1971) DOI; S. W. May et al., ibid. 99, 2017 (1977) DOI PubMed. Improved prepn: J. V. B. Kanth et al., Org. Process Res. Dev. 4, 550 (2000) DOI. Synthetic applications: H. C. Brown, S. K. Gupta, J. Am. Chem. Soc. 97, 5249 (1975) DOI; D. A. Evans, A. H. Hoveyda, J. Org. Chem. 55, 5190 (1990) DOI; D. A. Evans, G. C. Fu, ibid. 5678 DOI; D. J. Harrison et al., Tetrahedron Lett. 45, 8493 (2004) DOI; R. R. Huddleston et al., J. Org. Chem. 68, 11 (2003) DOI PubMed. Reviews: C. F. Lane, G. W. Kabalka, Tetrahedron 32, 981-990 (1976) DOI; G. W. Kabalka, Org. Prep. Proced. Int. 9, 133-147 (1977); B. Zhang, Synlett 2007, 666-667 DOI.