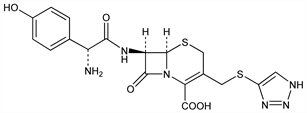
1915. Cefatrizine
Nomenclature
Description and references
Orally active semi-synthetic cephalosporin antibiotic. Prepn: G. L. Dunn, J. R. E. Hoover, DE 2316866; eidem, US 3867380 (1974, 1975 both to SK&F); D. Willner, L. B. Crast, DE 2364192 (1974 to Bristol-Myers), C.A. 81, 105538 (1974); G. L. Dunn et al., J. Antibiot. 29, 65 (1976). Prepn of the propylene glycolate: M. A. Kaplan, A. P. Granatek, DE 2500385; eidem, US 3970651 (1975, 1976 both to Bristol-Myers). In vitro and in vivo antibacterial activity and pharmacokinetic behavior: P. Actor et al., ibid. 28, 594 (1975); F. Leitner et al., Antimicrob. Agents Chemother. 7, 298 (1975). General pharmacological properties: M. Matsuzaki et al., Jpn. J. Antibiot. 29, 107 (1976), C.A. 85, 311g (1976). Determn in serum and urine by HPLC: E. Crombez et al., J. Chromatogr. 173, 165 (1979). Disposition of 14C-cefatrizine in man: R. C. Gaver, G. Deeb, Drug Metab. Dispos. 8, 157 (1980). Activity and clinical use: A. J. Weinstein, Drugs 20, 137 (1980). Acute toxicity: M. Matsuzaki et al., Jpn. J. Antibiot. 29, 612 (1976), C.A. 85, 171724y (1976). Toxicological studies: ibid. 639-686, C.A. 85, 171722w-171723x (1976). Series of articles on pharmacokinetics and clinical efficacy: Drugs Exp. Clin. Res. 11, 441-462 (1985).
Properties
Zwitterionic. LD50 in male, female mice, male, female rats (mg/kg): 6880, 6410, 4325, 4325 i.p. (Matsuzaki).Derivative
Compd with propylene glycol.Nomenclature
Properties
Rod-like crystals. [α]D23 +55.9° (c = 1% in 1N HCl). uv max: 227, 272 nm.Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Cephalosporins