1914. Cefamandole
Nomenclature
Description and references
Broad-spectrum semi-synthetic cephalosporin antibiotic. Prepn: C. W. Ryan, DE 2018600; idem, US 3641021 (1970, 1972 to Lilly); J. M. Greene, DE 2312997; idem, US 3840531 (1973, 1974 to Lilly). Biological properties: W. E. Wick, D. A. Preston, Antimicrob. Agents Chemother. 1, 221 (1972). Antibacterial activity: S. Eykyn et al., ibid. 3, 657 (1973); H. C. Neu, ibid. 6, 177 (1974); A. D. Russell, J. Antimicrob. Chemother. 1, 97 (1975). Pharmacologic studies: B. R. Meyers et al., Antimicrob. Agents Chemother. 9, 140 (1976); R. S. Griffith et al., ibid. 10, 814 (1976). Comprehensive description: R. H. Bishara, E. C. Rickard, Anal. Profiles Drug Subs. 9, 125-154 (1980).
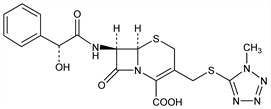
Derivative
Nafate.Nomenclature
Properties
White, odorless needles, mp 190° (dec). uv max (H2O): 269 nm (ε 10800). pKa 2.6-3.0. Sol in water, methanol. Practically insol in ether, chloroform, benzene, cyclohexane.Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Cephalosporins