1922. Cefepime
Nomenclature
Description and references
Semisynthetic, fourth generation cephalosporin antibiotic. Prepn: S. Aburaki et al., DE 3307550; eidem, US 4406899 (both 1983 to Bristol-Myers); and antibacterial activity: T. Naito et al., J. Antibiot. 39, 1092 (1986). In vitro comparative antimicrobial spectrum: N. J. Khan et al., Antimicrob. Agents Chemother. 26, 585 (1984); and β-lactamase stability: H. C. Neu et al., J. Antimicrob. Chemother. 17, 441 (1986). HPLC determn in plasma and urine: R. H. Barbhaiya et al., Antimicrob. Agents Chemother. 31, 55 (1987). Clinical evaluations in infection: N. Clynes et al., Diagn. Microbiol. Infect. Dis. 12, 257 (1989); S. Oster et al., Antimicrob. Agents Chemother. 34, 954 (1990). Review of clinical pharmacokinetics: M. P. Okamoto et al., Clin. Pharmacokinet. 25, 88-102 (1993).
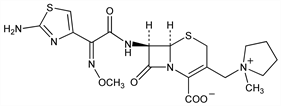
Properties
Colorless powder, mp 150° (dec). uv max (pH 7 phosphate buffer): 235, 257 nm (ε 16700, 16100).Derivative
Sulfate.Properties
mp 210° (dec). uv max (pH 7 phosphate buffer): 236, 258 nm (ε 17200, 16900).Derivative
Hydrochloride monohydrate.Nomenclature
Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Cephalosporins