1921. Cefditoren
Nomenclature
CAS number: 104145-95-1
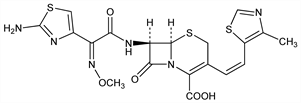
(6R,7R)-7-[[(2Z)-(2-Amino-4-thiazolyl)(methoxyimino)acetyl]amino]-3-[(1Z)-2-(4-methyl-5-thiazolyl)ethenyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic
acid; (+)-(6R,7R)-7-[2-(2-amino-4-thiazolyl)glyoxylamido]-3-[(Z)-2-(4-methyl-5-thiazolyl)vinyl]-8-oxo-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic
acid 72-(Z)-(O-methyloxime); ME-1206. C19H18N6O5S3; mol wt 506.58.
C 45.05%, H 3.58%, N 16.59%, O 15.79%, S 18.99%.
Description and references
Broad spectrum, third generation cephalosporin; active metabolite of orally absorbed pivaloyloxymethyl ester prodrug. Prepn: K. Atsumi et al., EP 175610; eidem, US 4839350 (1986, 1989 both to Meiji); K. Sakagami et al., J. Antibiot. 43, 1047 (1990); idem et al., Chem. Pharm. Bull. 39, 2433 (1991). In vitro and in vivo activities: A. Tamura et al., Antimicrob. Agents Chemother. 32, 1421 (1988). Extensive in vitro activity study: D. Felmingham et al., Drugs Exp. Clin. Res. 20, 127 (1994). Clinical pharmacokinetics and efficacy in pediatrics: N. Iwai et al., Jpn. J. Antibiot. 47, 181 (1994), C.A. 120, 260636e (1994).
Derivative
Sodium salt, sesquihydrate.C19H17N6NaO5S3.1 1/2 H2O; mol wt 537.57.
C 42.45%, H 3.37%, N 15.63%, Na 4.28%, O 16.37%, S 17.89%.
Properties
Pale yellow crystals from water, mp 195-200° (dec). [α]D20 +121.6° (c = 0.5 in methanol).Derivative
Pivaloyloxymethyl ester.Nomenclature
CAS number: 117467-28-4
Cefditoren pivoxil; ME-1207; Meiact (Meiji Seika). C25H28N6O7S3; mol wt 620.72.
C 48.37%, H 4.55%, N 13.54%, O 18.04%, S 15.50%.
Properties
Pale yellow powder, mp 127-129°. [α]D20 -48.5° (c = 0.5 in methanol).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Cephalosporins