1924. Cefixime
Nomenclature
Description and references
Orally active, third generation cephalosporin antibiotic. Prepn: T. Takaya et al., EP 30630; eidem, US 4409214 (1981, 1983 both to Fujisawa); H. Yamanaka et al., J. Antibiot. 38, 1738 (1985). Synthesis and activity of (E)-isomer: K. Kawabata et al., Chem. Pharm. Bull. 34, 3458 (1986). Mechanism of action: Y. Shigi et al., J. Antibiot. 37, 790 (1984). Comparative antibacterial spectrum in vitro and in vivo: T. Kamimura et al., Antimicrob. Agents Chemother. 25, 98 (1984). In vitro activity and β-lactamase stability: H. C. Neu et al., ibid. 26, 174 (1984). HPLC determn in human plasma and urine: Y. Tokuma et al., J. Chromatogr. 311, 339 (1984). Pharmacokinetics in humans: D. R. P. Guay et al., Antimicrob. Agents Chemother. 30, 485 (1986). Clinical trial in urinary tract infections: J. Levenstein et al., S. Afr. Med. J. 70, 455 (1986); A. Irvani et al., Am. J. Med. 85, Suppl. 3A, 17 (1988); in respiratory infections: R. Kiani et al., ibid. 6.
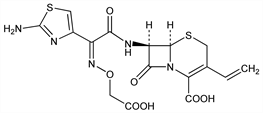
Derivative
(Z)-Form disodium salt.Properties
mp >250°.Derivative
(E)-Form trihydrate.Properties
Pale yellow solid, mp 218-225° (dec).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Cephalosporins