1925. Cefmenoxime
Nomenclature
Description and references
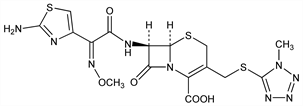
Third generation cephalosporin antibiotic; related structurally to cefotaxime and ceftizoxime, q.q.v. The name cefmenoxime applies to the isomer having a syn-methoxyimino group. Prepn (unspecified stereochemistry): M. Ochiai et al., DE 2556736; eidem, US 4098888 (1976, 1978 both to Takeda); syn-isomer: R. Heymes, A. Lutz, DE 2713272; eidem, US 4476122 (1983, 1984 both to Roussel-Uclaf). Series of articles on antibacterial activity, absorption, excretion, metabolism, mechanism of action, clinical studies: Chemotherapy (Tokyo) 29, Suppl. 1, 1-998 (1979). In vitro and in vivo study: K. Tsuchiya et al., Antimicrob. Agents Chemother. 19, 56 (1981). β-Lactamase stability: K. Okonogi et al., ibid. 20, 171 (1981). Clinical pharmacokinetics study: D. H"offler, P. Koeppe, Arzneim.-Forsch. 33, 269 (1983). Symposium: Am. J. Med. 77(6A), 1-59 (1984).
Derivative
Hydrochloride (syn-isomer).Nomenclature
Derivative
Sodium salt (syn-isomer).Nomenclature
Properties
[α]D20 -13.5 ±1° (c = 1 in water).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Cephalosporins