1929. Cefonicid
Nomenclature
Description and references
Injectable semi-synthetic cephalosporin antibiotic related to cefamandole, q.v. Prepn: D. A. Berges, DE 2611270; idem, US 4048311 (1976, 1977 both to Smith Kline); US 4093723, US 4159373 (1978, 1979 both to Smith Kline). Antibacterial activity, pharmacokinetics: P. Actor et al., Antimicrob. Agents Chemother. 13, 784 (1978). Series of articles on stability, comparative in vitro activity, serum levels: Curr. Chemother. Infect. Dis., Proc. 11th Int. Congr. Chemother. vol. 1 (Am. Soc. Microbiol., Washington, D.C., 1979) pp 246-254. Stability towards β-lactamases: R. Mehta et al., J. Antibiot. 34, 202 (1981). Kinetics and renal handling: D. Pitkin et al., Clin. Pharmacol. Ther. 30, 587 (1981). In vitro evaluation vs Group B streptococci: A. S. Bayer et al., Antimicrob. Agents Chemother. 21, 344 (1982). Review of antibacterial activity, pharmacology, therapeutic use: E. Saltiel, R. N. Brogden, Drugs 32, 222-259 (1986).
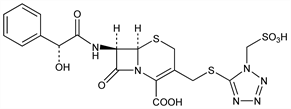
Derivative
Disodium salt.Nomenclature
Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Cephalosporins