1946. Ceftazidime
Nomenclature
Description and references
Third generation cephalosporin antibiotic. Prepn: C. H. O'Callaghan et al., DE 2921316; eidem, US 4258041 (1979, 1981 both to Glaxo). Prepn of crystalline pentahydrate: A. Brodie, L. A. Wetherill, DE 3037102; eidem, US 4329453 (1981, 1982 both to Glaxo). Chemical and antibacterial properties: eidem, Antimicrob. Agents Chemother. 17, 876 (1980). Activity vs Pseudomonas and Enterobacteriaceae: L. Verbist, J. Verhaegen, ibid. 807. In vitro comparison with other β-lactams: R. Wise et al., ibid. 884; L. Verbist, ibid. 19, 407 (1981). Symposium on clinical studies: J. Antimicrob. Chemother. 12, Suppl. A, 1-414 (1983). Comprehensive description: M. A. Abounassif et al., Anal. Profiles Drug Subs. 19, 95-121 (1990). Review of antibacterial activity, pharmacokinetics and therapeutic use: C. P. Rains et al., Drugs 49, 577-617 (1995).
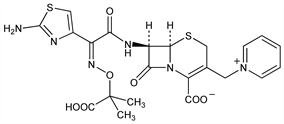
Derivative
Pentahydrate.Nomenclature
Properties
Crystalline solid. uv max (pH 6): 257 nm (E1%1cm 348). Sol in alkali, DMSO; slightly sol in DMF, methanol, water. Insol in acetone, alcohol, chloroform, dioxane, ether, ethyl acetate, toluene.Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial, esp in reptiles.Keywords
Antibacterial (Antibiotics); β-Lactams; Cephalosporins