10378. Ceftaroline
Nomenclature
Description and references
Fourth generation cephalosporin; active form of the N-phosphono prodrug, ceftaroline fosamil. Prepn: H. Tawada, K. Okonogi, JP 9100283 (1997 to Takeda); of N-phosphono prodrug: T. Ishikawa et al., WO 9932497; eidem, US 6417175 (1999, 2002 both to Takeda). Synthesis and physiochemical properties: T. Ishikawa et al., Bioorg. Med. Chem. 11, 2427 (2003) DOI PubMed. Stability study: Y. Ikeda et al., Chem. Pharm. Bull. 56, 1406 (2008) DOI PubMed. Comparative antibacterial spectrum in vitro: Y. Iizawa et al., J. Infect. Chemother. 10, 146 (2004) DOI PubMed. Experimental pharmacokinetics and pharmacodynamics: D. Andes, W. A. Craig, Antimicrob. Agents Chemother. 50, 1376 (2006) DOI PubMed. Comparative in vivo activity in experimental methicillin resistant Staphylococcus aureus (MRSA) infection: C. Jacqueline et al., ibid. 51, 3397 (2007) DOI PubMed. Clinical comparison with standard therapy for complicated skin and skin structure infections: G. H. Talbot et al., ibid. 51, 3612 (2007) DOI PubMed. Comprehensive review: D. Parish, N. Scheinfeld, Curr. Opin. Investig. Drugs 9, 201-209 (2008) PubMed.
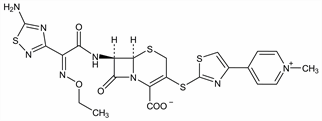
Properties
Soly in water: 2.3 mg/ml.Derivative
Ceftaroline fosamil.Nomenclature
Properties
Amorphous solid. Cryst from aq acetic acid as the monoacetic acid solvate, mp 221-223° (dec). Soly in water at 25°: >100 mg/ml.Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Cephalosporins