1952. Ceftobiprole Medocaril
Nomenclature
Description and references
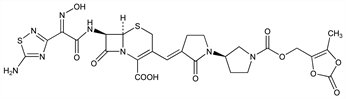
Fourth generation cephalosporin antibiotic prodrug; rapidly cleaved in plasma to the active metabolite, ceftobiprole. Prepn: P. Hebeisen et al., WO 9965920; eidem, US 6232306 (1999, 2001 both to Hoffmann-La Roche). In vitro and in vivo activity against methicillin-resistant Staphylococcus aureus (MRSA): idem et al., Antimicrob. Agents Chemother. 45, 825 (2001). Comparative antibacterial spectrum in vitro: R. N. Jones et al., J. Antimicrob. Chemother. 50, 915 (2002). In vivo activity in experimental pneumococcal pneumonia: E. Azoulay-Dupuis et al., Antimicrob. Agents Chemother. 48, 1105 (2004). Clinical pharmacokinetics and safety: A. Schmitt-Hoffmann et al., ibid. 48, 2576 (2004).
Derivative
Sodium salt.Nomenclature
Properties
Sol in water.Derivative
Ceftobiprole.Nomenclature
Description and references
Prepn: P. Angehrn et al., EP 849269; eidem, US 5981519 (1997, 1999 both to Hoffmann-La Roche).
Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Cephalosporins