1953. Ceftriaxone
Nomenclature
Description and references
Parenteral third generation cephalosporin antibiotic. Prepn: M. Montavon, R. Reiner, GB 2022090; eidem, US 4327210 (1979, 1982 both to Hoffmann-La Roche); R. Reiner et al., J. Antibiot. 33, 783 (1980). In vitro and in vivo studies: P. Angehrn et al., Antimicrob. Agents Chemother. 18, 913 (1980). Pharmacokinetics: M. Seddon et al., ibid. 240. Determn in plasma, urine, bile: K. H. Trautmann, P. Haefelfinger, J. High Resolut. Chromatogr. Chromatogr. Commun. 4, 54 (1981), C.A. 94, 202437 (1981). Mechanism of action study: R. B. Wright et al., J. Antibiot. 34, 590 (1981). Kinetics in humans: K. Stoekel et al., Clin. Pharmacol. Ther. 29, 650 (1981). Toxicity data: K. Teelman et al., “Experimentelle Toxikologie von Ceftriaxon”, in Ceftriaxon ein neues parenterales Cephalosporin, Proc. Hahnenklee Symp., 1981, R. Grieshalber, Ed. (Editiones Roche, Basel, 1982) pp 91-111. Review of antibacterial activity, toxicology, pharmacology and clinical studies: D. M. Richards et al., Drugs 27, 469 (1984); T. R. Beam, Pharmacotherapy 5, 237-253 (1985).
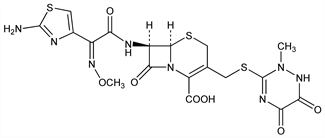
Derivative
Disodium salt hemiheptahydrate.Nomenclature
Properties
White cryst powder, mp >155° (dec). [α]D25 -165° (c = 1 in water) (calc for water-free substance). uv max (water): 242, 272 nm (ε 32300, 29530). pKa: ≈3 (COOH), 3.2 (NH3+), 4.1 (enolic OH). Soly in water at 25°: ≈40 g/100 ml. LD50 in male, female, mice, rats (mg/kg): 3000, 2800, 2175, 2175 i.v.; >10000 all species orally; >5000 all species s.c. (Teelman).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Cephalosporins