1954. Cefuroxime
Nomenclature
Description and references
Prepn: M. C. Cook et al., DE 2439880; eidem, US 3974153 (1973, 1976 both to Glaxo). Prepn of the 1-acetoxyethyl ester: M. Gregson, B. Sykes, DE 2706413; eidem, US 4267320 (1977, 1981 both to Glaxo). In vitro studies: C. H. O'Callaghan et al., Antimicrob. Agents Chemother. 9, 511 (1976); R. N. Jones et al., ibid. 12, 47 (1977). In vitro antibacterial activity, human pharmacokinetics: C. H. O'Callaghan et al., J. Antibiot. 29, 29 (1976). Pharmacology: H. Freiesleben et al., Proc. 10th Int. Congr. Chemother., Zürich, 1977 (Am. Soc. for Microbiol., Washington, 1978) II, pp 873-874. Pharmacokinetics: P. E. Gower, ibid. 877-878; J. Kosmidis et al., ibid. 875-876. Clinical studies: P. F. Wood et al., ibid. 1042-1044; R. Norrby et al., J. Antimicrob. Chemother. 3, 355 (1977). Review of antibacterial activity, pharmacology and therapeutic efficacy: R. N. Brogden et al., Drugs 17, 233-266 (1979). Comprehensive description: T. J. Wozniak, J. R. Hicks, Anal. Profiles Drug Subs. 20, 209-236 (1991).
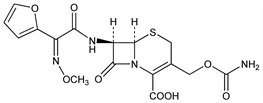
Properties
White crystalline solid. [α]D20 +63.7° (c = 1.0 in 0.2M pH 7 phosphate buffer). uv max (pH 6 phosphate buffer): 274 nm (ε 17600).Derivative
Sodium salt.Nomenclature
Properties
White solid. [α]D20 +60° (c = 0.91 in water). uv max (water): 274 nm (ε 17400). Freely sol in water and buffered solutions; sol in methanol; very slightly sol in ethyl acetate, diethyl ether, octanol, benzene and chloroform. Soly in water: 500 mg/2.5 ml. pKa (water): 2.5; (DMF): 5.1. Solns are stable at room temp for 13 hrs; <10% decompn in 48 hrs at 25° (O'Callaghan, J. Antibiot.).Derivative
1-Acetoxyethyl ester.Nomenclature
Cefuroxime axetil; CCI-15641; Ceftin (GSK); Cefurax (Betapharm); Cepazine (Sanofi-Synthelabo); Elobact (Cascan); Oraxim (Menarini); Zinat (GSK); Zinnat (GSK).Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); β-Lactams; Cephalosporins