1960. Celiprolol
Nomenclature
Description and references
Cardioselective β1-adrenergic blocker. Prepn: BE 823411; G. Z"olss et al., US 4034009 (1975, 1977 both to Chemie Linz). Hemodynamic effects: J. Bonelli et al., Wien. Klin. Wochenschr. 90, 350 (1978); H. Pittner, Arch. Pharmacol. 311, Suppl., 180 (1980). Series of articles on determn in biological material, pharmacology, toxicology, clinical studies: Arzneim.-Forsch. 33, 1-79 (1983). Toxicity: W. Wendtlandt, H. Pittner, ibid. 41. Symposium on pharmacology, clinical efficacy and comparison with other β-blockers: J. Cardiovasc. Pharmacol. 8, Suppl. 4, S1-S152 (1986). Comprehensive description: D. J. Mazzo et al., Anal. Profiles Drug Subs. 20, 237-301 (1991).
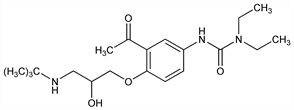
Properties
Crystals, mp 110-112°.Derivative
Hydrochloride.Nomenclature
Properties
White, odorless crystals, mp 197-200° (dec). Soly at ≈25°C (g/100 ml): water 15.1, methanol 18.2, ethanol 1.61, chloroform 0.42. uv max (water): 231, 324 nm (E1% 652, 57); (0.01N HCl): 231, 324 nm (E1% 660, 60); (0.01N NaOH): 231, 324 nm (E1% 640, 60); (methanol): 232, 329 nm (E1% 775, 58). LD50 in male mice, rats (mg/kg): 56.2, 68.3 i.v.; 1834, 3826 orally (Wendtlandt, Pittner).Therapeutic Category
Antihypertensive, antianginal.
Keywords
β-Adrenergic Blocker; Antianginal; Antihypertensive; Aryloxypropanolamine Derivatives