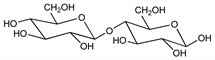
1961. Cellobiose
Nomenclature
Description and references
Unit of cellulose and lichenin. Does not occur free in nature, or as glucoside. Prepn from cotton: Braun, Org. Synth. coll. vol. II, 122, 124 (1943). Prepn from cell-free enzymatic hydrolyzate of cellulose: Whistler, Smart, J. Am. Chem. Soc. 75, 1916 (1953). Structure: Haworth, Hirst, J. Chem. Soc. 119, 193 (1923); Charlton et al., ibid. 1926, 89; Zemplén, Ber. 59, 1254 (1926); Haworth et al., J. Chem. Soc. 1927, 2809; Peterson, Spencer, J. Am. Chem. Soc. 49, 2822 (1927); Helferich et al., Ber. 63, 992 (1930); Hess, Dziengel, ibid. 68, 1594 (1935); Hassid, Ballou in The Carbohydrates, W. Pigman, Ed. (Academic Press, New York, 1957) p 490. Synthesis: Haskins et al., J. Am. Chem. Soc. 64, 1289 (1942). Review: Pazur in The Carbohydrates vol. 2A, W. Pigman et al., Eds. (Academic Press, New York, 2nd ed., 1970) pp 109-110; R. G. Edwards, Dev. Food Carbohydr. 2, 229-273 (1980).