1984. Cephapirin Sodium
Nomenclature
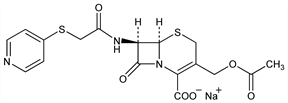
CAS number: 24356-60-3
(6R,7R)-3-[(Acetyloxy)methyl]-8-oxo-7-[[(4-pyridinylthio)acetyl]amino]-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic
acid monosodium salt; 3-(hydroxymethyl)-8-oxo-7-[2-(4-pyridylthio)acetamido]-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic
acid acetate monosodium salt; 7-[α-(4-pyridylthio)acetamido]cephalosporanic acid sodium salt; sodium 7-(pyrid-4-ylthioacetamido)cephalosporanate; BL-P-1322; Ambrocef (Lusofarmaco); Brisfirina (BMS); Bristocef (BMS); Cefadyl (BMS); Cefa-Lak (Fort Dodge); Piricef (CT); ToDay (Franklin). C17H16N3NaO6S2; mol wt 445.45.
C 45.84%, H 3.62%, N 9.43%, Na 5.16%, O 21.55%, S 14.40%.
Description and references
A cephalosporin C antibiotic effective against gram-pos. and gram-neg. bacteria including Staphylococcus aureus, Escherichia coli, Klebsiella pneumoniae and Proteus mirabilis. Prepn: L. B. Crast, ZA 6707783; eidem, US 3422100 (1968, 1970 both to Bristol-Myers). Alternate processes: H. H. Silvestri, D. A. Johnson, US 3503967; R. E. Havranek, L. B. Crast, US 3578661 (1970, 1971 both to Bristol-Myers); L. B. Crast et al., J. Med. Chem. 16, 1413 (1973). Activity studies: Chisholm et al., Antimicrob. Agents Chemother. 1969, 244; Axelrod et al., Appl. Microbiol. 22, 904 (1971); Bran et al., Antimicrob. Agents Chemother. 1, 35 (1972); Wiesner et al., ibid. 303.
Properties
White crystalline powder, sol in water.Therapeutic Category
Antibacterial.
Therapeutic Category (Veterinary)
Antibacterial.Keywords
Antibacterial (Antibiotics); β-Lactams; Cephalosporins