2022. Cetirizine
Nomenclature
Description and references
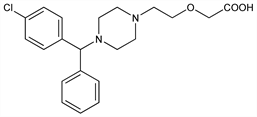
Nonsedating type histamine H1-receptor antagonist; major metabolite of hydroxyzine, q.v. Pharmacological activity resides primarily in the (R)-isomer. Prepn: E. Baltes et al., EP 58146; eidem, US 4525358 (1982, 1985 both to UCB); of enantiomers: E. Cossement et al., GB 2225321 (1989 to UCB). See also: eidem, US 5478941 (1995 to UCB). Enantioselective HPLC determn in urine: S. O. Choi et al., Arch. Pharmacal Res. 23, 178 (2000). Stereoselective binding study: M. Gillard et al., Mol. Pharmacol. 61, 391 (2002). Clinical pharmacokinetics: E. Baltes et al., Fundam. Clin. Pharmacol. 15, 269 (2001). Clinical comparison with enantiomers on cutaneous response: J. L. Devalia et al., Allergy 56, 50 (2001); on nasal response: D. Y. Wang et al., ibid. 339. Review of pharmacology and clinical experience in allergic disorders: J. M. Portnoy, C. Dinakar, Expert Opin. Pharmacother. 5, 125-135 (2004).
Properties
Crystals from ethanol, mp 110-115°.Derivative
Dihydrochloride.Nomenclature
Properties
Crystals from isopropanol, mp 225°. Sol in water.Derivative
(R)-Form.Nomenclature
Derivative
(R)-Form dihydrochloride.Nomenclature
Properties
mp 229.3°. [α]D25 -12.79° (c = 1 in water).Therapeutic Category
Antihistaminic.
Keywords
Antihistaminic; Piperazines