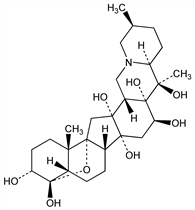
2035. Cevine
Nomenclature
CAS number: 124-98-1
(3α,4α,16β)-4,9-Epoxycevane-3,4,12,14,16,17,20-heptol. C27H43NO8; mol wt 509.63.
C 63.63%, H 8.50%, N 2.75%, O 25.12%.
Description and references
By hydrolysis of cevadine. Structure and stereochemistry: Barton et al., Experientia 10, 81 (1954); Kupchan et al., J. Am. Chem. Soc. 80, 1769 (1958); Kupchan et al., Tetrahedron 7, 47 (1959); Eeles, Tetrahedron Letters no. 7, 24 (1960). Comparative toxicity: O. Krayer et al., J. Pharmacol. Exp. Ther. 82, 167 (1944); K. Tanaka, ibid. 113, 89 (1955).