13-01932. Cefclidin [Archived]
Nomenclature
Description and references
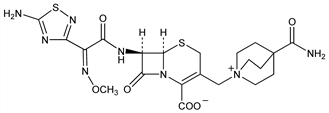
Broad spectrum fourth generation cephalosporin antibiotic. Prepn: H. Yamauchi et al., EP 188255; eidem, US 4748171 (1986, 1988 both to Eisai). Structure activity study: I. Sugiyama et al., J. Antibiot. 45, 103 (1992). Comparative in vitro antibacterial spectrum and β-lactamase resistance: N.-A. Watanabe et al., Antimicrob. Agents Chemother. 32, 693 (1988). In vivo antipseudomonal activity: T. Toyosawa et al., Chemotherapy (Tokyo) 42, 1242 (1994). Clinical pharmacokinetics: M. Nakashima et al., J. Clin. Pharmacol. 29, 144 (1989). Toxicity study: K. Ogura et al., Chemotherapy (Tokyo) 40, Suppl. 4, 95 (1992).
Properties
LD50 in male, female mice, rats (mg/kg): >10000, >10000, 2236, 2147 i.v.; >10000 both species orally; >10000 both species s.c.; >5000 both species i.m. (Ogura).Therapeutic Category
Antibacterial.
Keywords
Antibacterial (Antibiotics); β-Lactams; Cephalosporins
Status
This monograph has been retired and is no longer subject to revision or update.