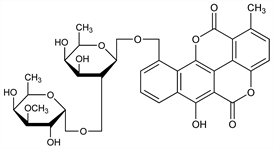
2045. Chartreusin
Nomenclature
CAS number: 6377-18-0
10-[[6-Deoxy-2-O-(6-deoxy-3-O-methyl-α-d-galactopyranosyl)-β-d-galactopyranosyl]oxy]-6-hydroxy-1-methylbenzo[h][1]benzopyrano[5,4,3-cde][1]benzopyran-5,12-dione; antibiotic X-465A; lambdamycin; NSC-5159. C32H32O14; mol wt 640.59.
C 60.00%, H 5.04%, O 34.97%.
Description and references
Antibiotic substance produced by Streptomyces chartreusis from African soil, also by another Streptomyces sp. from North American soil: B. E. Leach et al., J. Am. Chem. Soc. 75, 4011 (1953). Identity with Antibiotic X-465A: J. Berger et al., ibid. 80, 1636 (1958). Structure: L. H. Sternbach et al., ibid. 1639; E. Simonitsch et al., Helv. Chim. Acta 47, 1459 (1964); W. Eisenhuth et al., ibid. 1475. Anticancer activity: J. P. McGovern et al., Cancer Res. 37, 1666 (1977). Synthesis of chartarin, the chartreusin aglycone: T. R. Kelly et al., J. Am. Chem. Soc. 102, 798 (1980); F. M. Hauser, D.W. Combs, J. Org. Chem. 45, 4071 (1980).