2071. Chloral Hydrate
Nomenclature
CAS number: 302-17-0
2,2,2-Trichloro-1,1-ethanediol; trichloroacetaldehyde monohydrate; Escre (SS Pharm.); Noctec (BMS); Nycton; Somnos (Merck & Co.); Chloraldurat (Pohl). C2H3Cl3O2; mol wt 165.40.
C 14.52%, H 1.83%, Cl 64.30%, O 19.35%.
Description and references
Made by adding the required amount of water to trichloroacetaldehyde, q.v. First synthesized by Liebig in 1832. Introduced as hypnotic by Liebreich in 1869. Comprehensive description: J. E. Fairbrother, Anal. Profiles Drug Subs. 2, 85-143 (1973). Toxicity data: E. Goldenthal, Toxicol. Appl. Pharmacol. 18, 185 (1971). Evaluation of mutagenicity: J. Leuschner, F. Leuschner, Arzneim.-Forsch. 41, 1101 (1991).
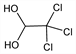
Derivative
Monohydrate.Properties
Large, monoclinic plates. Perfect 001 cleavage. Skin irritant. Aromatic, penetrating and sightly acrid odor, slightly bitter, caustic taste. Slowly volatilizes on exposure to air. d 1.91. mp 57° when heated in an open vessel. bp 98° with dissociation into chloral and water. Freely sol in water. One ml of water dissolves the following amounts of chloral hydrate: 2.4 g at 0°, 8.3 g at 25°, 14.3 g at 40°. One gram of chlorate hydrate dissolves in 1.3 ml alcohol, in 2 ml chloroform, in 1.5 ml ether, in 1.4 ml olive oil, in 0.5 glycerol, in 68 g carbon disulfide. Freely sol in acetone, methyl ethyl ketone. Moderately or sparingly sol in turpentine, petr ether, carbon tetrachloride, benzene, toluene. Dissociation in water: Piguet et al., Helv. Chim. Acta 46, 406 (1963). An alcoholic soln of chloral hydrate (1 in 20) does not at once redden moistened blue litmus paper. Dec by NaOH into chloroform. Reduces ammoniacal AgNO3. Liquefies when triturated with an equal quantity of menthol, camphor or thymol. LD50 orally in rats: 479 mg/kg (Goldenthal).Note
This is a controlled substance (depressant): 21 CFR, 1308.14.Use
Manuf DDT.Therapeutic Category
Sedative, hypnotic.
Therapeutic Category (Veterinary)
Anesthetic, sedative, hypnotic.Keywords
Sedative/Hypnotic; Chloral Derivatives