2081. Chlorcyclizine
Nomenclature
CAS number: 82-93-9
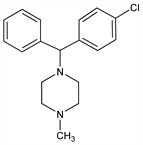
1-[(4-Chlorophenyl)phenylmethyl]-4-methylpiperazine; 1-(p-chloro-α-phenylbenzyl)-4-methylpiperazine; 1-(4-chlorobenzhydryl)-4-methylpiperazine; N-methyl-N′-(4-chlorobenzhydryl)piperazine; chlorocyclizine; compd 47-282; Alergicide; Perazyl; Trihistan. C18H21ClN2; mol wt 300.83.
C 71.87%, H 7.04%, Cl 11.79%, N 9.31%.
Description and references
Prepn: Baltzly et al., J. Org. Chem. 14, 775 (1949); Murfitt, Dewing, GB 656043 (1951 to Wellcome Found.); US 2630435 (1953 to Burroughs Wellcome). Toxicity study: Castillo et al., J. Pharmacol. Exp. Ther. 96, 388 (1949).
Properties
Oil, bp0.1-0.15 137-145°.Derivative
Hydrochloride.Nomenclature
CAS number: 1620-21-9
AH-289; Di-Paralene (Abbott); Perazil (Burroughs Wellcome); Histantin (Burroughs Wellcome). C18H21ClN2.HCl; mol wt 337.29.
C 64.10%, H 6.57%, Cl 21.02%, N 8.31%.
Properties
Crystalline powder, mp 226-227°. One gram dissolves in about 2 ml water, in 11 ml alc, in about 4 ml chloroform. Practically insol in ether, benzene.Derivative
Dihydrochloride.C18H21ClN2.2HCl; mol wt 373.75.
C 57.84%, H 6.20%, Cl 28.46%, N 7.50%.
Properties
Prisms from alc + ether, mp 216-216.5°. Freely sol in water; sol in alc. Aq soln is acid to litmus. LD50 i.p. in mice: 137 mg/kg (Castillo).Derivative
Di[(tert-butyl)naphthalene sodium sulfonate].Nomenclature
Bexedan (Smit).C18H21ClN2.2C14H15NaOS; mol wt 809.47.
C 68.25%, H 6.35%, Cl 4.38%, N 3.46%, Na 5.68%, O 3.95%, S 7.92%.
Therapeutic Category
Antihistaminic.
Therapeutic Category (Veterinary)
The hydrochloride as antihistaminic.Keywords
Antihistaminic; Piperazines