2108. Chlornaphazine
Nomenclature
CAS number: 494-03-1
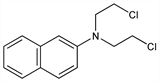
N,N-Bis(2-chloroethyl)-2-naphthylamine; dichloroethyl-β-naphthylamine; β-naphthyldi(2-chloroethyl)amine; β-naphthylbis(β-chloroethyl)amine; di(2-chloroethyl)-β-naphthylamine; CB-1048; R-48; Cloronaftina; Erysan. C14H15Cl2N; mol wt 268.18.
C 62.70%, H 5.64%, Cl 26.44%, N 5.22%.
Description and references
Prepd from 2-C10H7N(C2H4OH)2 by treatment with POCl3: Ross, J. Chem. Soc. 1949, 183; Ghielmetti, Farmaco Ed. Sci. 5, 275 (1950); 11, 603 (1956). Mechanism of action studies: Jeney et al., Kiserl. Orvostud. 20, 369 (1968), C.A. 70, 18766j (1969). Review of carcinogenicity studies: IARC Monographs 4, 119-124 (1974).
Properties
Platelets from petr ether, mp 54-56°. bp5 210°. Very sparingly sol in water, glycerol. More sol (in ascending degree) in petr ether, ethanol, olive oil, ether, acetone, benzene.Caution
This substance has been listed as a known carcinogen: Fourth Annual Report on Carcinogens (NTP 85-002, 1985) p 44; delisted because no U.S. residents exposed: Fifth Annual Report on Carcinogens (NTP 89-239, 1989) p 340.Therapeutic Category
Antineoplastic.
Keywords
Antineoplastic; Alkylating Agents; Nitrogen Mustards