C
8H
8ClNO; mol wt 169.61.
C 56.65%, H 4.75%, Cl 20.90%, N 8.26%, O 9.43%.
Description and references
Prepn of m-, o- and p-isomers: Roberts et al., J. Org.
Chem. 24, 654 (1959). Sepn of o- and p-isomers: Orton, Bradford, J. Chem. Soc. 1927, 986.
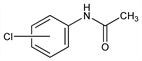
Derivative
m-Chloroacetanilide.
Nomenclature
N-(3-Chlorophenyl)acetamide; 3′-chloroacetanilide. Properties
Needles from 50% glacial acetic acid, mp 77-78°. Readily sol
in alcohol, benzene, carbon disulfide; very slightly sol in ligroin.
uv max (95% ethanol): 245 nm (log ε 4.19).Derivative
o-Chloroacetanilide.
Properties
Needles from dil glacial acetic acid, mp 87-88°. Sublimes
at about 50-60°. Practically insol in water, alkalies; sol in alc;
more sol in benzene than corresponding p-isomer. uv max (95% ethanol): 240 nm (log ε 4.02).Derivative
p-Chloroacetanilide.
Properties
Orthorhombic crystals from aq glacial acetic acid,
alc, or acetone. mp 178-179°. d422 1.385. Practically insol in water; readily sol in alc,
ether, carbon disulfide; slightly sol in CCl4, benzene.
uv max (95% ethanol): 249 nm (log ε 4.25).